Probiotic dietary Intervention in Polycystic Ovary Syndrome – A Randomised Controlled Trial
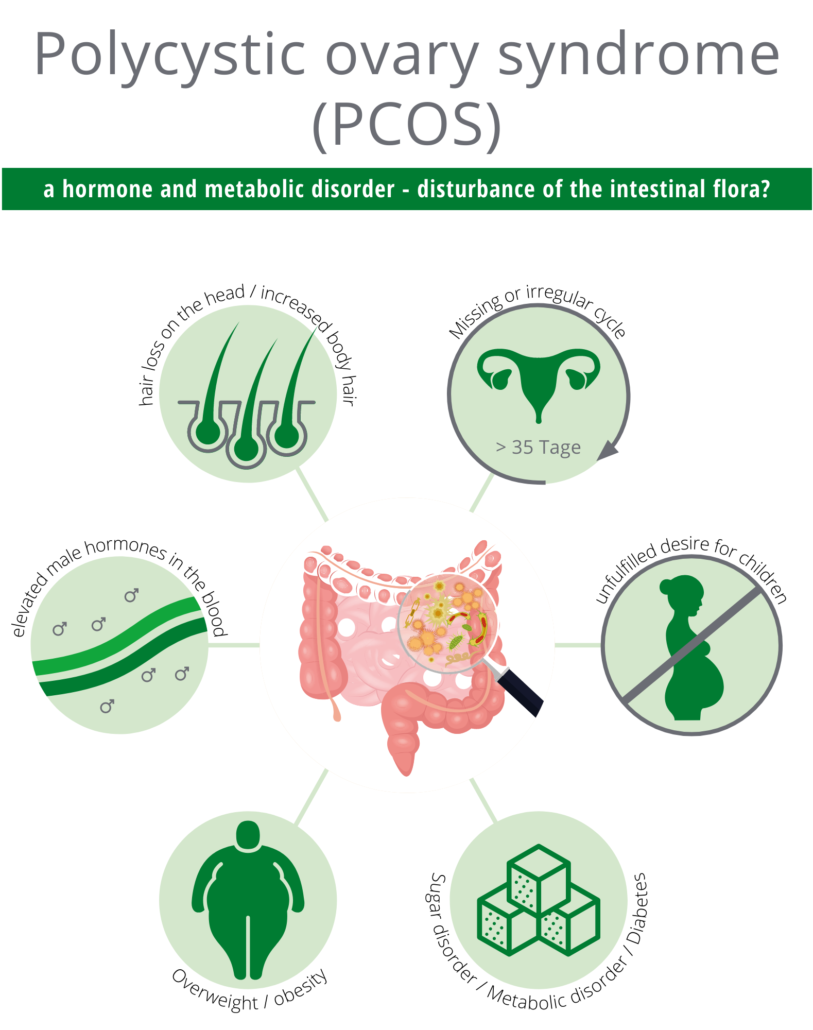
Polycystic Ovary Syndrome (PCOS) is a hormonal and metabolic disorder caused by elevated androgen levels in women. A variety of symptoms are common, including irregular/missing menstrual cycles, increased body hair growth or parietal hair loss, unfulfilled child wish, weight gain/obesity and dysfunctional glucose metabolism, predisposing to type 2 diabetes mellitus.
These symptoms are most commonly treated either by contraceptive pills or the insulin sensitizer metformin with the potential of severe side effects. Therefore, we aim for new, non-hormonal therapeutic options.
We recently documented altered gut bacteria composition and reduced gut microbiome diversity in PCOS women (Lindheim et al. 2017, Haudum et al. 2020). Some of these bacteria have a significant impact on hormonal and metabolic regulation making probiotics a potential treatment option for women with PCOS.
The PRO-PCO RCT (randomized controlled trial) is a clinical intervention in cooperation with Institute Allergosan, including 180 women with PCOS either treated with probiotic, placebo (to test the effectiveness of the probiotics) or metformin (to compare the probiotics to a standard therapy) for six months.
Participants attend three visits at our endocrine outpatients’ department at the University Hospital of Graz. Detailed hormonal and metabolic blood analyses including an oral glucose tolerance test, questionnaires and microbiome sampling are assessed. Stool and urine samples are collected at home for microbiome and gut permeability tests. Study medication (probiotics/placebo/metformin) is then taken for six months, before returning for the final visit, where the assessments are repeated and further treatment is advised.
The aim of this study is a comprehensive validation of viable alternatives to already existing treatment options for PCOS women, supporting their individual health and quality of life.