Tracer Imaging and In-Vivo Imaging at CBmed
Nuclear medicine that uses radioactive tracers (radiopharmaceuticals) to assess bodily functions is one of CBmed’s core labs. One project aims to develop small-molecule based radio tracers for in-vivo PET imaging of immune checkpoint axis and test them in preclinical systems.
Vienna is not only the home of the second branch of CBmed – it is also the origin of a technology developed already in the early 20th century. Tracer imaging can be seen as a speciality in modern medicine, but its foundations have been discovered early by Hungarian radiochemist George Charles de Hevesy. Together with Frederic Paneth, he carried out the first radioactive-tracer experiment 1913 at the Vienna Institute of Radium Research, leading to the development of radioactive tracers to study chemical processes such as in the metabolism of animals. Hevesy not only performed the first radiotracer studies on plants and animals, using both natural and artificial radionuclides, he also performed the first tracer studies employing stable nuclides by using deuterated water to measure the turnover of hydrogen in the body. His groundbreaking, scientific efforts have been recognized in 1943 with the Nobel Prize in Chemistry laureate.
In his early beginnings, de Hevesy conceived the radiotracer technique by which radioisotopes could be used to investigate the behaviors of stable atoms. It is a technique second to none in its analytical power, and its principles remain basically the same up to this day: Modern nuclear medicine uses radioactive tracers (or radiopharmaceuticals) to assess bodily functions, diagnose and treat disease. With specially designed devices and microscopes, it is possible to track the path of these radioactive tracers. To put it simple, this method allows to follow the way of radioactivity through a living organism without intervening into the system itself. The radioactive substances in question are being used in extremely low dosage, which means that they have no medical effect – but they provide a deeper and more complete view into organisms and vital systems.
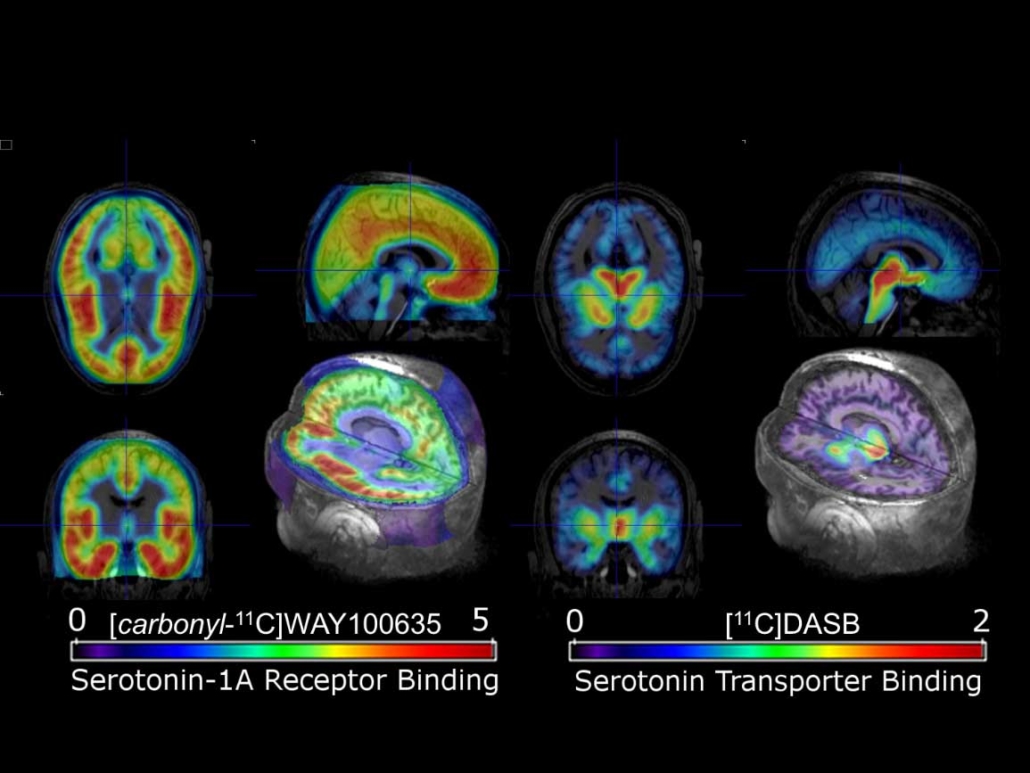
PET-Imaging using specific radiopharmaceuticals targeting molecular sub-structures in the brain associated with serotonin-mediated neurotransmission.
Credits/source: Rupert Lanzenberger, Medical University of Vienna.
Tracer imaging in modern nuclear medicine
For most diagnostic studies in modern nuclear medicine, the radioactive tracer is given to the patient through an intravenous injection. However, the tracer can also be administered by inhalation, an oral ingestion or by a direct injection into a specific organ. The two most common imaging modalities in nuclear medicine are „Single Photon Emission Computed Tomography“ or SPECT and „Positron Emission Tomography“ or PET scans. SPECT imaging instruments provide three-dimensional tomographic images of the distribution of radioactive tracer molecules that have been introduced into the patients body. CBmed focusses on PET scans, that use radiopharmaceuticals to create three-dimensional images as well. The main difference is the type of radiotracers used: SPECT scans measure gamma rays, while the decay of the radio tracers used with PET scans produce small particles – positrons. These positrons are particles with roughly the same mass as an electron, but oppositely charged. Scanning small amounts of energy, the detectors in the PET scanner measure photons and use this information to create images of internal organs.
In theory, PET offers countless opportunities, because each substance can be marked with a radioactive substance, given some technical limitations. However, it’s a cost-intense procedure, and after all, it deals with radioactive, ionizing radiation, which is regulated by laws and legal limitations. So in practice, PET scans are used to detect cancer and monitor its progression, response to treatment, and to detect metastases. The majority of the applications is in the field of oncology, often they are used for staging, to examine the extent of the cancer, such as how large the tumor is, and if it has spread. This can have a direct effect on the therapy decision since the PET images show only living cells – cells with metabolism that are able to absorb the substance in question. A dead cell which has been damaged by the effects of a therapy therefore cannot be made visible by this technique. Besides oncology, PET is often used in (nuclear) cardiology, experimental applications and in clinical studies, particularly in psychiatry and neurology.
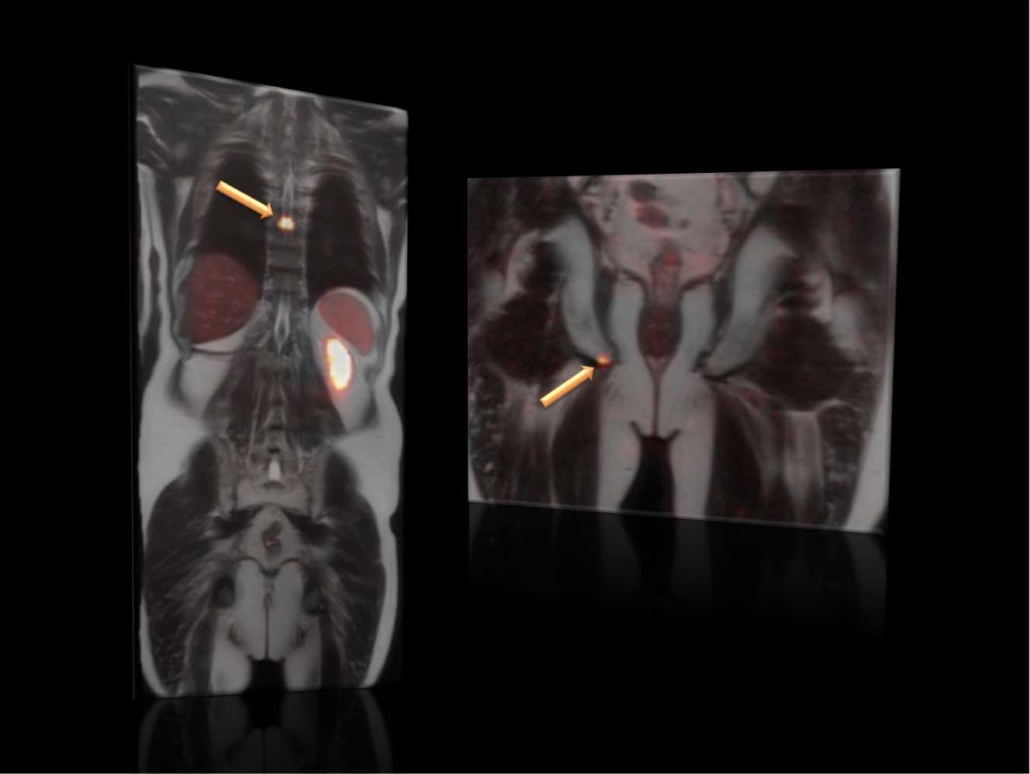
Combined PET/MRI-Imaging using [68Ga]Ga-PSMA-11 to visualize metastases (arrows) from prostate cancer by molecular targeting the prostate specific membrane antigen (PSMA).
Credits/source: Markus Hartenbach, Medical University of Vienna.
Current tracer imaging research at CBmed
At CBmed, tracer and in-vivo imaging is one of the core-technologies, and the core-lab is open to all kinds of research and scientific questions. So far, the focus has been on oncological questions, diabetes or immune checkpoint inhibitors. Following the tracer technology was a logical choice, since specific tracers can be seen as a personalized treatment with a direct read-out of scanned objects. A big advantage is the formal set-up of CBmeds second scientific base in Vienna: One of the scientific partners is the Medical University of Vienna, which runs its own cyclotron – a particle accelerator which can be used for the production of the radioactive substances. The second scientific partner is the Veterinary Medical University of Vienna, which runs a Small Animal or Pre-Clinical Imaging facility. Besides the scientific partners, there are also two medical companies on board: ITM (Isotopen Technologie München) from Germany and SOFIE Biosciences from Los Angeles. The latter produces an automated radiochemical synthesizer for the production of preclinical and clinical PET probes. Through the interaction of all those different partners, it is possible to deliver a full package, completed by CBmed’s Digital Pathology, that can produce videos based on proteins, which has an influence on metabolites, metabolomics, genetics and genomics.
Currently, CBmed’s most ambitions project concerning tracer technologies is „Project 2.21 – Translation Molecular Imaging Agents For In-Vitro And In-Vivo Application“. The aim is to develop small-molecule based radio tracers for in-vivo PET imaging of immune checkpoint axis and test them in preclinical systems. The radiotracer development uses organic and medicinal chemistry, radiochemistry using different radionuclides, automation techniques and in-vitro and in-vivo assays. IHC (Immunohistochemistry) is set in place for the evaluation of tumor evolution, dissemination or metastases, infiltration into surrounding tissue and the involvement of stroll cells. Despite an interruption due to the Corona Lockdown, the project is well on track, and results are expected for the end of 2021/beginning of 2022.
The Author
Assoc.-Prof. Wolfgang Wadsak, MSc. PhD
Head of Medicinal Radiochemistry and Biomarker Development
Department of Biomedical Imaging and Image-guided Therapy (Division of Nuclear Medicine)



 Pixabay
Pixabay